(ITUNES OR LISTEN HERE)
The Free Open Access Medical Education (FOAM)
We review Dr. Rory Spiegel’s, A Case of Identity Part Two, post on EMNerd which is essentially a take down of dual antiplatelet therapy (DAT) in acute coronary syndrome (ACS). The bottom line? There’s no demonstrable and clinically significant benefit from DAT as demonstrated in the trials below.
CURE trial – composite endpoints of questionable clinical significance and an enormous sample size.
- 2.1% absolute decrease in cardiovascular death and myocardial infarction (MI), completely powered by the 1.5% absolute difference in MIs. Almost all of these MIs were Type IV and peri-procedural. Mortality between groups was identical at 30 days and end of follow up (1.0% vs 1.1% and 2.3% vs 2.4%, respectively).
ACCOAST – RCT of prasugrel or placebo prior to angiography
- No difference in cardiovascular death, myocardial infarct, stroke, urgent revascularization or glycoprotein IIb/IIIa rescue therapy (10.8% vs 10.8%)
- Approximately 1% increase in major bleeding
CREDO – RCT with placebo or clopidogrel 3-24 hours prior to urgent cardiac catheterization
- No statistical difference in the rates of death, stroke or MI at 28 days
- Statistical significance of a secondary endpoint of the 1-year outcomes with a 2% absolute reduction in the rate of death, MI, and stroke, largely the result of a 1.9% reduction of MIs.
- 1% increase in major bleeding events
- In patients with non-ST elevation ACS, pretreatment with thienopyridines is not associated with reduced mortality but comes at a cost of a significant excess of major bleeding.
Composite endpoints are problematic (see this post, “Would You Rather“) and statistical significance claimed in these trials is largely a product of composite outcomes rather than patient oriented measures.
The Bread and Butter
We summarize some key topics from the following readings, Goldfrank (10th ed) Chapter 60, EMPractice October 2013 (there’s almost nothing in Rosenalli on this topic) but, the point isn’t to just take our word for it. Go enrich your fundamental understanding yourself!
Aspirin
- Irreversibly inhibits platelets (for the duration of platelet’s life)
Ibuprofen
- Reversibly inhibits platelets
Novel Oral Anticoagulants (NOACs)
NOACs have gained increased popularity and are slowly supplanting warfarin for common anticoagulation indications such as non-valvular atrial fibrillation (NVAF) as well as treatment of venous thromboembolisms such as pulmonary embolism (PE) and deep venous thrombosis (DVT).
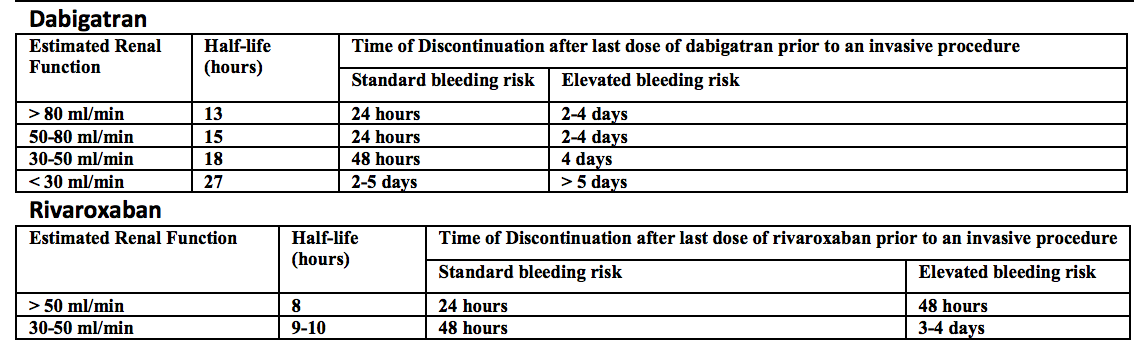
Direct thrombin inhibitor – dabigatran (Pradaxa). This drug was the first to supplant warfarin for NVAF in the United States, billed as more patient friendly given the lack of purported need for routine monitoring. Recent investigations by Cohen et al, however, demonstrate that monitoring may, in fact, be safer. Further, in a real world, retrospective cohort of Medicare beneficiaries given either dabigatran or warfarin for atrial fibrillation, major bleeding of the dabigatran cohort was higher than in the warfarin cohort 9.0% (95% CI 7.8 – 10.2) versus 5.9% (95% CI 5.1 – 6.6) after propensity matching [Hernandez]. For more on the problems with dabigatran, check out Emergency Medicine Literature of Note.
- Predominantly renal excretion
- Caution with impaired renal function (can cause dabigatran to stick around longer)
- Hemodialysis an option in acute overdose; however, most people would probably not want to put a dialysis catheter in a coagulopathic patient.
- Half-life ~ 15 hours
- Can elevate the PTT. If the PTT is normal, likely not coagulopathic secondary to dabigatran [Dager et al].
- No reversal agent
Factor XA inhibitors – these have XA in the name….rivaroXAban, apiXAban, edoXAban.
Rivaroxaban – approved for NVAF and treatment of DVT/PE. Half life approximately 6-9 hours.
Apixaban – approved for NVAF and treatment of DVT/PE. Half life about 12 hours.
Edoxaban – approved for NVAF. Half life about 10-14 hours.
- Cleared by liver and kidneys.
- Can elevate the prothrombin time (PT), but not reliably. Specific assays exist but are not widely available and are expensive.
- No specific reversal agent although andexanet alfa is in the pipeline. It’s a Factor Xa decoy (Andexanet Alfa) that binds up the F10A inhibitors like a sponge. Read more here.
- In the setting of major bleeding, guidelines recommend 4 factor PCCs. A recent study demonstrates reduction in bleeding using 4 factor PCCs on healthy patients given edoxaban [Zahir et al, EMLitofNote]. The benefit of 4 factor PCCs is predominantly based on improvement in numbers, not patient oriented benefit and is discussed in these posts by Dr. Spiegel The Sign of Four, The Sign of Four Part 2.
More FOAM on Anticoagulation Reversal
Generously Donated Rosh Review Questions
Question 1. A 65-year-old man with a metal aortic valve presents with hematemesis. His vitals are BP 95/50 and HR 118. The patient is on warfarin and has an INR of 7.3. [polldaddy poll=8501352]
Question 2. A 66-year-old woman with atrial fibrillation on warfarin presents with dark stools for 2 days. Her vitals are T 37.7°C, HR 136, BP 81/43, RR 24, and oxygen saturation 94%. Her labs reveal a hematocrit of 19.4% (baseline 33.1%) and an INR of 6.1. [polldaddy poll=8505511]
Answers.
1. D. The patient presents with life-threatening bleeding and an elevated INR from warfarin use requiring immediate anticoagulant reversal regardless of the indication for anticoagulation. Warfarin acts by inhibiting vitamin K recycling thus limiting the effectiveness of vitamin K dependant clotting factors (factors II, VII, IX and X). The effect of warfarin can be measured using the prothrombin time or the INR. Warfarin is indicated for anticoagulation for a number of disorders including the presence of a metal valve. Patients with metal valves are at a higher 1-year risk of clot formation around the valve and subsequent embolic stroke. The therapeutic goal of warfarin in a patient with a metallic valve is usually between 2.5 – 3.5 or 3.0 – 4.0. Despite the increased stroke risk, patients with life-threatening bleeding should always have their warfarin reversed by administration of vitamin K and fresh frozen plasma (FFP). Alternatively, prothrombin complex concentrates can be given instead of FFP.
Warfarin is not amenable to hemodialysis (A) for removal or reversal. Although patients with a mechanical valve are at an increased stroke risk (increased 1 year risk) reversal should not be delayed (B), as the patient is more likely to die in the immediate situation from their gastrointestinal bleed. Platelet transfusion (C) will not help, as warfarin does not inhibit platelet function.
2. C. The patient has a life-threatening gastrointestinal bleed in the setting of anticoagulation with warfarin, a vitamin K antagonist. Warfarin acts by inhibiting the synthesis of vitamin K-dependant factors in the coagulation cascade (II, VII, IX, X, protein C, and protein S). The anticoagulant effect of warfarin should be reversed as part of the patient’s emergent treatment. Fresh frozen plasma (FFP) contains all factors in the coagulation cascade and should be given in patients with major bleeding and elevated INR. Vitamin K should be given IV in critically ill patients with elevated INR because it shortens the time to effect.
Vitamin K should not be given intramuscularly (B) because absorption via this route is highly variable. Vitamin K should also not be given orally (D) in critically ill patients because the onset of action will be delayed. Additionally, absorption in patients with gastrointestinal bleeding may be variable. Vitamin K should be given along with FFP (A) because the factors inhibited rely on vitamin K for function.