We cover a post by Dr. Rory Spiegel, EMNerd: The Case of Differing Perspectives, on the results of the ATACH-2 trial on blood pressure control in intracranial hemorrhage (ICH). This study sought to determine the safety and efficacy of the intensive blood pressure lowering.
Population: adults (>18 y/o) with ICH on CT scan, GCS ≥ 5 and <4.5 hours since symptom onset (changed mid-study)
Intervention: Reduce and maintain the hourly minimum systolic blood pressure in the range of 110 to 139 mm Hg throughout the period of 24 hours after randomization (“intensive treatment”). Preferred agents for blood pressure control in order of preference were 1. nicardipine 2. labetalol (diltiazem or urapidil if not available)
- Mean minimum in hours 0-2: 128.9±16 mm Hg
Control: Reduce and maintain the hourly minimum systolic blood pressure in the range of 140 to 179 mm Hg throughout the period of 24 hours after randomization.
- Mean minimum in hours 0-2: 141.1±14.8 mm Hg
Outcome: The primary outcome was the proportion of patients who had moderately severe or severe disability (modified Rankin scale score (mRS) 4-5) or those who had died (mRS 6; hereafter referred to as “death or disability”) at 3 months.
- death or disability: Intensive treatment = 186 participants (38.7%) vs Standard treatment = 181 (37.7%)
Bottom Line: INTENSIVE blood pressure lowering <140mmHg is not supported by the literature. AHA and Rosens/Tintinalli recommend goal of 160-180 mmHg or diastolic <130 mmHg.
We also discuss the history of blood pressure control in ICH and the pendulum swing on this in recent years, using an episode of the Skeptic’s Guide to Emergency Medicine, Episode 73. This episode covers the Interact-2 trial.
Core Content
We delve into core content on ICH using Rosen’s (8th ed) and Tintinalli (8th ed)
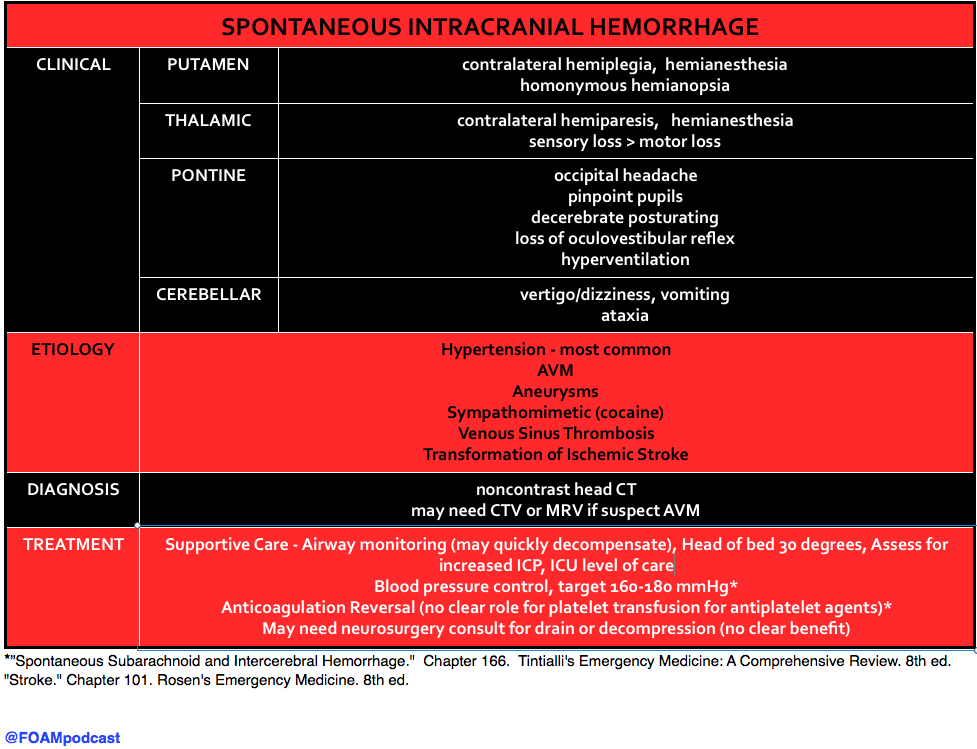
The use of platelets in patients with ICH on antiplatelet agents (aspirin, clopidogrel, etc) has been controversial and both Rosen’s and Tintinalli acknowledge that there is no good quality evidence that platelets are beneficial in this population. The PATCH trial, recently published in the Lancet, found no benefit to platelet transfusion in patients with ICH who had taken antiplatelet agents in the 7 days prior to their ICH. See this review from Dr. Salim Rezaie of Rebel EM.
Population: patients ≥18years of age with non-traumatic ICH confirmed by CT and GCS ≥ 8 who took antiplatelet agent for at least 7 days prior (excluded thrombocytopenia/known coagulopathy)
Intervention: platelet transfusion
Control: standard care without platelet confusion
Outcome: primary outcome was the difference in functional outcome at 3 months after randomisation scored with the mRS
- Odds of a shift towards death or dependence at 3 months: adjusted OR 2.05 (95% CI 1·18–3·56; p = 0.0114). This favors the control group.
- Secondary outcome of survival: 68% in platelet transfusion group vs 77% in standard group (p = 0.15)
Note: Like many stroke trials, the planned statistical analysis plan changed after the data came in. Initially authors planned a fixed dichotomous analysis (yes or no, did they have an mRS of 4-6 at 3 months). They changed this to an ordinal logistic regression analysis of the shift of all categories of the mRS at 3 months as this has greater “statistical efficiency”.
Generously Donated Rosh Review Questions
Question 1. A patient presents with nausea, vomiting, right-sided hemiplegia and non-occipital headache. His gaze is deviated to the left, but he denies loss of sensation. Thirty minutes later, he becomes stuporous and progresses into coma. The pupils are now fixed and dilated. Abnormal posturing is absent. A brain CT scan is ordered. You would expect to find intracerebral hemorrhage in which of the following sites
A. Left pons
B. Left putamen
C. Right cerebellum
D. Right thalamus
[accordion]
[toggle title=”Answer” state=”closed”]
A large putamen hemorrhage results in a rapid progression of hemiplegia, nausea, vomiting and headache over 30 minutes, which is quickly followed by ipsilateral deviation of the eyes,stupor, coma and mydriatic pupils (“blown-pupil”, associated with brainstem compression and occulomotor nerve palsy). Acute management includes controlling intracranial pressure and hypertension. Surgical removal of clots is seldom successful, however, some neurologic function may be salvaged in those without coma or those with lobar clots.
Pontine hemorrhages (A) are characterized by total paralysis, rapid coma, decerebrate rigidity (abnormal posturing marked by shoulder adduction, elbow extension, wrist pronation and flexion, digit flexion, leg extension and plantarflexion) and small (miotic), not mydriatic, reactive pupils. Cerebellar insults (C) are evidenced by vomiting, occipital headache, inability to stand, vertigo, and eye deviation to the opposite side of the lesion. In this scenario, the eye deviation makes sense, but not the presenting symptoms.Thalamic lesions (D) are marked by complete hemisensory loss. Also common is hemiparesis (bleeding extension into the internal capsule) and aphasia.
Putamen Hemorrhage: Contralateral hemiparesis/hemiplegia, Contralateral sensory loss, Homonymous hemianopia
[/toggle]
[/accordion]
References:
- Qureshi AI, Palesch YY, Barsan WG, et al. Intensive Blood-Pressure Lowering in Patients with Acute Cerebral Hemorrhage. N Engl J Med [Internet] 2016;NEJMoa1603460. Available from: http://www.nejm.org/doi/10.1056/NEJMoa1603460
- Anderson C, Heeley E, Huang Y, et al. Rapid Blood-Pressure Lowering in Patients with Acute Intracerebral Hemorrhage. N Engl J Med [Internet] 2013;368(25):2355–65. Available from: http://www.nejm.org/doi/abs/10.1056/NEJMoa1214609
- “Spontaneous Subarachnoid and Intercerebral Hemorrhage.” Chapter 166. Tintialli’s Emergency Medicine: A Comprehensive Review. 8th ed.
- “Stroke.” Chapter 101. Rosen’s Emergency Medicine. 8th ed.
- Baharoglu MI, Cordonnier C, Salman RA-S, et al. Platelet transfusion versus standard care after acute stroke due to spontaneous cerebral haemorrhage associated with antiplatelet therapy (PATCH): a randomised, open-label, phase 3 trial. Lancet [Internet] 2016;6736(16):1–9. Available from: http://linkinghub.elsevier.com/retrieve/pii/S0140673616303920