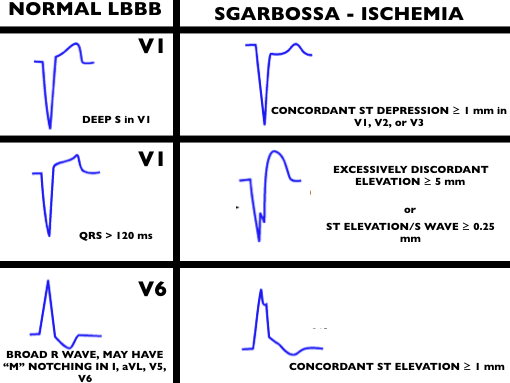
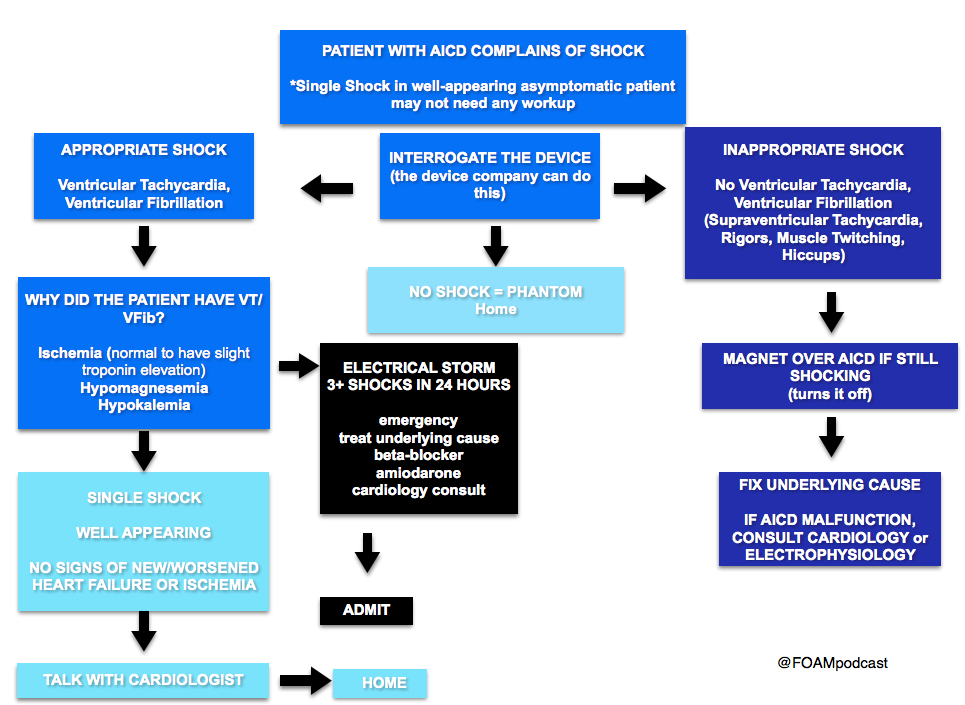
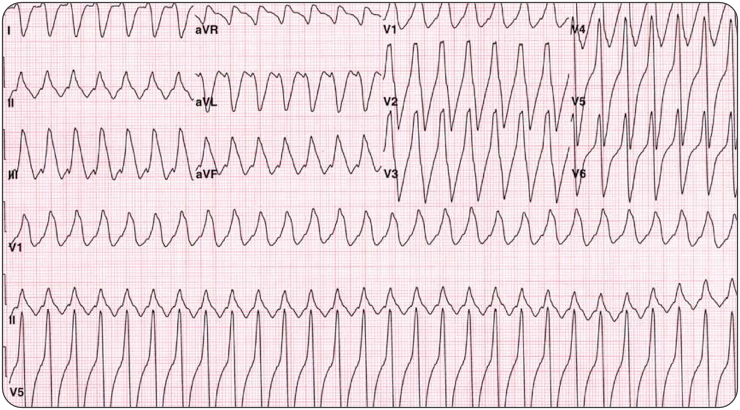
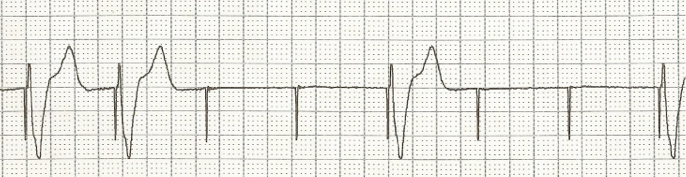
(ITUNES OR LISTEN HERE)
The Free Open Access Medical Education (FOAM)
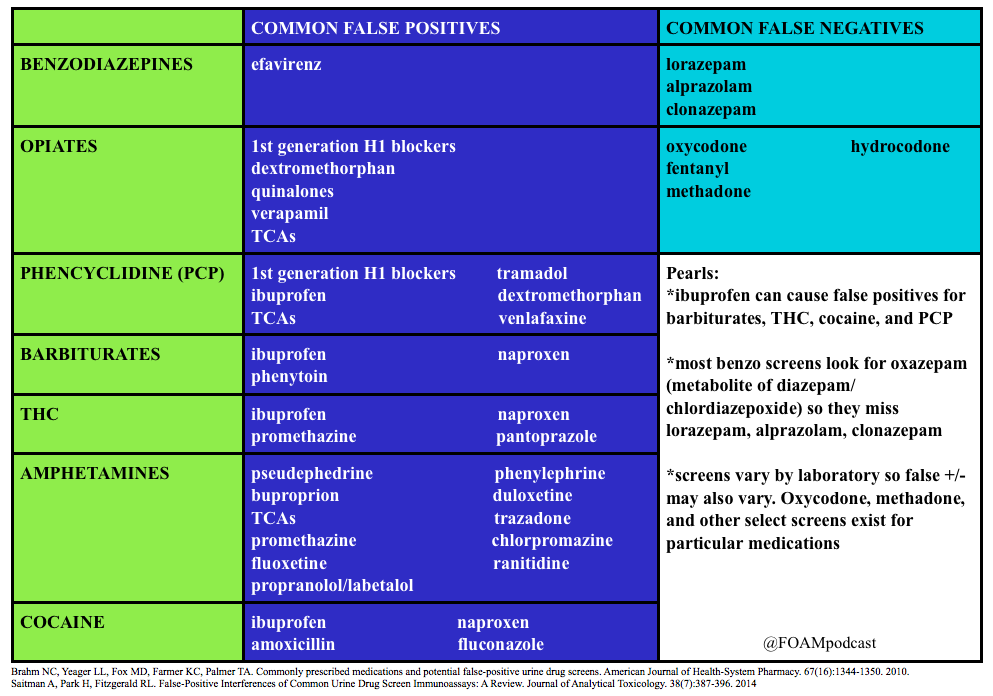
We review a post by Dr. Seth Trueger (@MDaware) on false positives of common medications in the urine drug screen. We delve into posts by Dr. Bryan Hayes (@PharmERToxGuy) on false negatives for benzodiazepines and opioids in the urine drug screen.
Core Content
We delve into core content on cocaine and phencyclidine (PCP) using Rosen’s (8th edition), Chapter 154 and Tintinalli (8th edition)
Cocaine
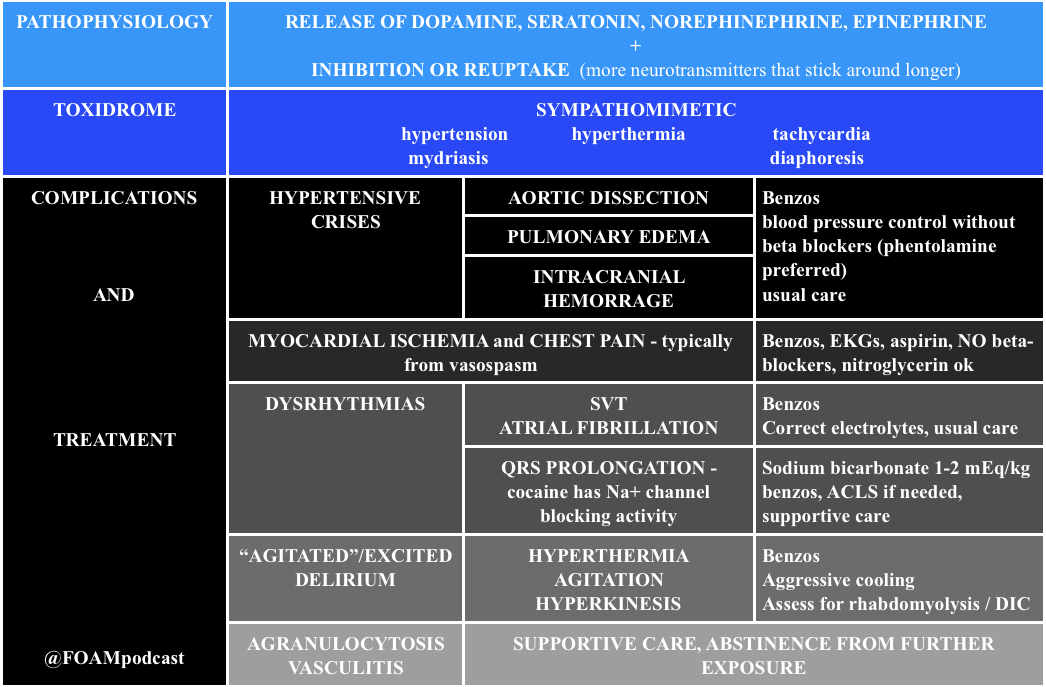
Note: Beta-blockers are contra-indicated in cocaine induced hypertension and chest pain. Much of the ischemia induced by cocaine is thought to be due to vasospasm, predominantly from alpha-1 receptor effects. Beta-blockers block the relaxation provided by beta-2 stimulus on muscles, leaving alpha-1 constricting vessels, “unopposed.” This is largely theoretical/based on canine literature as there are only two human studies on this, (a) 10 humans given propranolol + cocaine with vasospasm and (b) 9 humans given labetalol + cocaine without vasospasm [4,5]. However, recent papers attempting to dispel this teaching don’t quite prove the point. One retrospective chart review looked at cocaine positive urine screens in patients with chest pain and found no worsened troponins. Cocaine stays positive in the urine for 3 days so it is not clear that these were patients presenting with cocaine associated chest pain [6].
Generously Donated Rosh Review Questions
- An 18-year-old man is brought to the ED by the police after being found running around a parking lot, screaming at bystanders. He reportedly smoked phencyclidine (PCP) earlier that day. His vital signs are notable for a heart rate of 130 beats per minute and a blood pressure of 150/86 mm Hg. On physical exam, he is diaphoretic, with vertical nystagmus and equal pupils and appears acutely agitated.
A. Administer haloperidol for acute agitation
B. Administer IV fluids for presumed rhabdomyolysis and benzodiazepines for agitation
C. Apply physical restraints; this is adequate treatment for most cases
D. Monitor for traumatic injuries; they are the most common cause of morbidity and mortality with PCP use
E. Urinary acidification to increase PCP elimination
[accordion]
[toggle title=”Answer” state=”closed”]
The most common cause of morbidity and mortality in patients with phencyclidine (PCP) intoxication is rhabdomyolysis. Fluid hydration with normal saline is the initial therapy for rhabdomyolysis. Creatinine kinase (CK) should be obtained in patients with PCP intoxication. If abnormal, serial levels should be obtained until a downward trend is noted. Serum creatinine should also be monitored for evidence of renal insufficiency. PCP is a dissociative anesthetic drug that works on the NMDA glutamate receptor. People who abuse PCP can have sensations of euphoria initially, but this can be followed by an emergence reaction that is characterized by dysphoria and psychosis. If initial calming measures such as placing the patient in a calm environment fail and chemical restraints are needed, benzodiazepines are the treatment of choice. Benzodiazepines are also the preferred medication for acute agitation. Haloperidol (A) may cause dysrhythmias and hypotension. It also lowers the seizure threshold and may precipitate PCP-induced movement disorders. PCP is a weak base and its excretion is increased in acidic urine. Physical restraints (C) may be required for patients with PCP intoxication, but they should be used in conjunction with chemical restraints (preferably benzodiazepines). An agitated patient fighting against physical restraints is at risk for worsening rhabdomyolysis, which, if unrecognized, can precipitate cardiac arrest. Traumatic injuries (D)frequently occur in PCP-intoxicated patients; however, rhabdomyolysis with subsequent renal insufficiency is the most common cause of associated morbidity and mortality. Patients with PCP intoxication should be evaluated for suspected trauma, and any injuries should be stabilized and treated. Acidification of the urine (E) can cause a metabolic acidosis, which is potentially harmful and has not demonstrated improved outcomes. For these reasons, urinary acidification is not routinely recommended.
[/toggle]
[/accordion]
2. A 39-year-old woman presents with chest pain and difficulty breathing that began shortly after smoking crack cocaine. Vital signs are BP 190/85 mm Hg, HR 105, RR 18, T 99.1℉. The ECG demonstrates ST segment depression and T wave inversions in the lateral leads.
A. Diazepam
B. Metoprolol
C. Morphine
D. Phenylephrine
[accordion]
[toggle title=”Answer” state=”closed”]
Diazepam, a benzodiazepine, should be administered to this patient who is hyperadrenergic from cocaine intoxication. Signs and symptoms of cocaine intoxication include, dilated pupils, diaphoresis, tachycardia, hypertension and hyperthermia. Many patients experience euphoria, though some will develop acute psychosis. Benzodiazepines decrease the cocaine-induced hyperadrenergic state. Reduction of sympathetic tone induces coronary and peripheral vasodilation. Coronary artery dilation directly improves myocardial blood flow. Peripheral vasodilation reduces preload and afterload. Reductions in preload and afterload decrease blood pressure and improve myocardial oxygen demand. Several factors, including, excess sympathetic stimulation, dehydration, hyperthermia, and cocaine-induced cardiac sodium channel blockade, may cause patients with cocaine intoxication to develop dysrhythmias. These contributing factors should be treated with benzodiazepines, IV fluid resuscitation and temperature management. In some patients, cocaine-induced cardiac sodium channel blockade may cause wide complex tachycardia that should be treated with sodium bicarbonate. Hyperthermia should be managed aggressively with a target temperature of less than or equal to 102.0℉. Severe agitation, aggression or psychosis should be initially managed with benzodiazepines. Most antipsychotic agents have pronounced anticholinergic side effects. This may worsen dysrhythmias.and decrease sweating, further complicating temperature management
[/toggle]
[/accordion]
- Rao R, Hoffman RS. Cocaine and other Sympathomimetics. Rosen’s Emergency Medicine (8e). Chapter 154, 1999-2006.e2
- “Cocaine and Amphetamines.” Tintinalli’s Emergency Medicine: A Comprehensive Study Guide (8e). Chapter 187
- “Prison Medicine.” Tintinalli’s Emergency Medicine: A Comprehensive Study Guide (8e). Chapter 301
- Lange RA, Cigarroa RG, Flores ED, et al. Potentiation of cocaine, induced coronary vasoconstriction by beta adrenergic blockade. Ann Intern Med. 1990;112:897–90
- Boehrer JD, Moliterno DJ, Willard JE, Hillis LD, Lange RA. Influence of labetalol on cocaine-induced coronary vasoconstriction in humans. Am J Med. 1993;94(6):608–610
- Ibrahim M, Maselli DJ, Hasan R, Hamilton A. Safety of β-blockers in the acute management of cocaine-associated chest pain. The American Journal of Emergency Medicine. 31(3):613-616.