Apple Podcasts , Spotify, Listen Here
The American Academy of Pediatrics (AAP) released new clinical practice guidelines in August 2021 on the evaluation and management of well-appearing febrile infants ages 8-60 days old (Pantell et al).



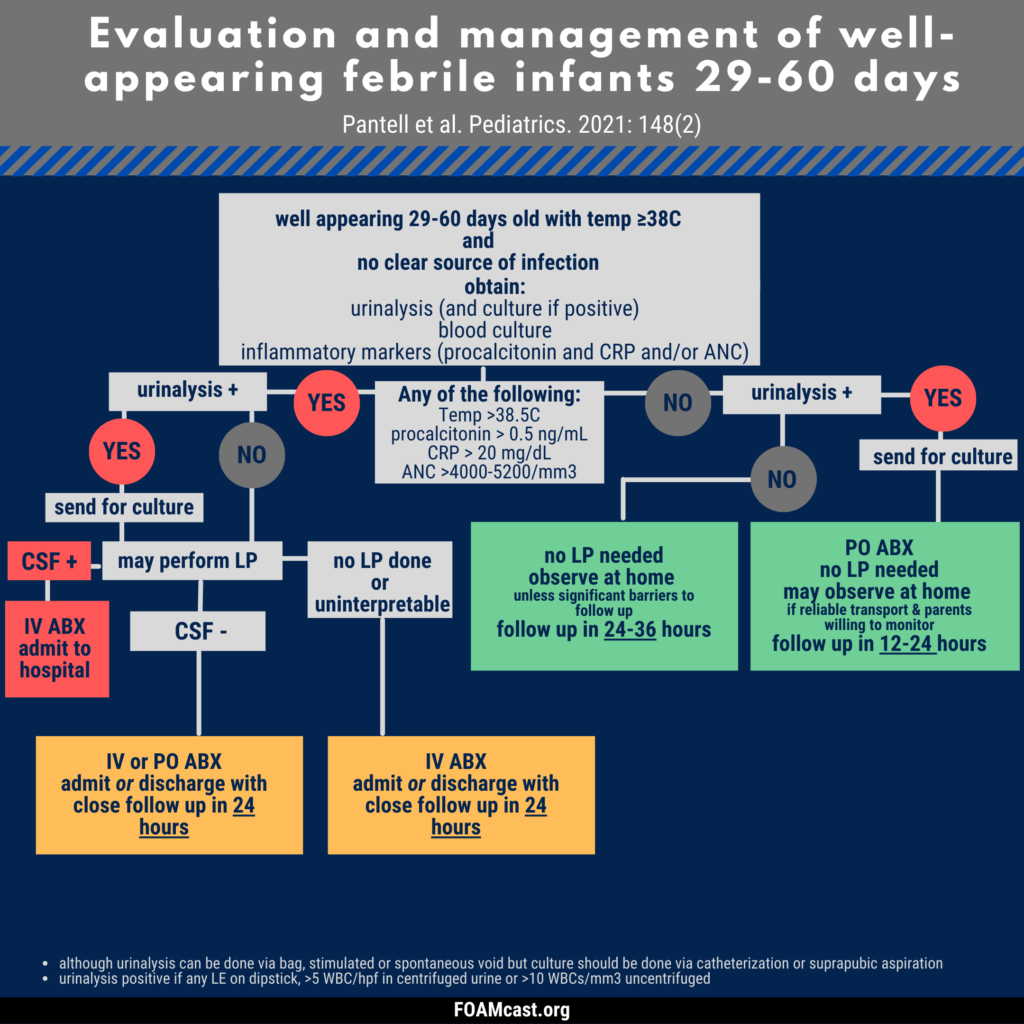
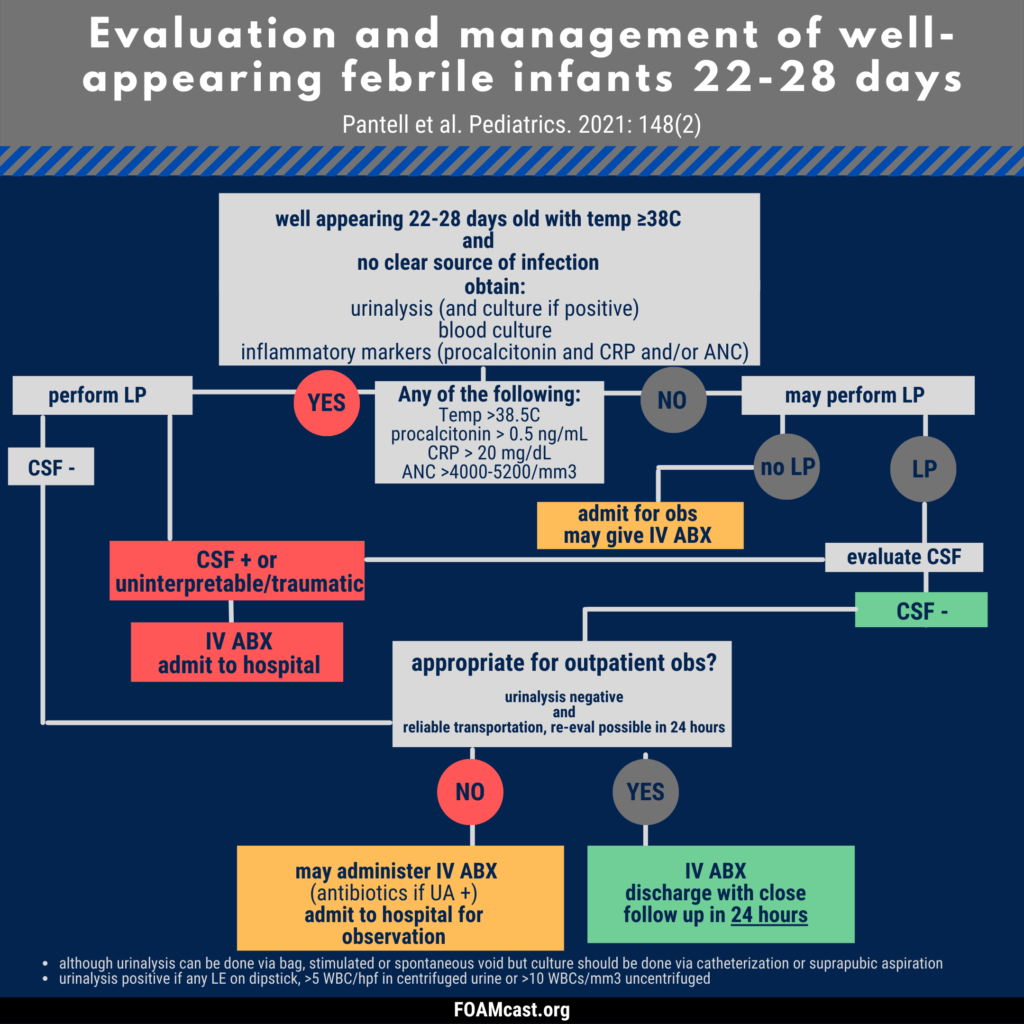
Apple Podcasts , Spotify, Listen Here
The American Academy of Pediatrics (AAP) released new clinical practice guidelines in August 2021 on the evaluation and management of well-appearing febrile infants ages 8-60 days old (Pantell et al).





Apple Podcasts , Spotify, Listen Here
On September 24, 2021, the United States’ CDC made recommendations on who should or may receive a COVID-19 vaccine booster. These recommendations overstepped the vote made by the CDC’s Advisory Committee on Immunization Practices (ACIP), who voted against the recommendation that those 18-64 who have occupational exposure may receive a booster vaccine. In this podcast, we discuss the current evidence as of September 24, 2021 on boosters as well as the evidence-based risk-benefit analysis from the CDC ACIP meeting. Jeremy’s insights can be found at Inside Medicine.
First, the data presented from Pfizer is minimal and involves a total of 312 individuals who received boosters. We have no insight into effectiveness based on this.

In Israel, boosters were available to those ≥60 who completed the series ≥5 months prior beginning at the end of July. Bar-On et al report on this natural experiment.


The data presented at the CDC ACIP meeting can be found in the presentation slides available here.


Apple Podcasts , Spotify, Listen Here
In this podcast, we review pulmonary embolism (PE) risk stratification and dive into the use of right ventricular dysfunction as a predictor of mortality. We highlight a patient-level meta-analysis by Becattini et al and discuss guidelines on PE risk stratification.




Apple Podcasts , Spotify, Listen Here
In this podcast, we summarize a practice-changing article of 2021 that addresses awareness during paralysis, Pappal et al in Annals of Emergency Medicine.


Apple Podcasts , Spotify, Listen Here
We review a recent paper by Kilaru et al on pharmacy fills by ED patients for naloxone after an opioid-related ED visit. We also discuss harm reduction and the reasons this is important. For more on harm reduction, including patient education materials, see this.



updated September 23, 2021
We have several podcasts on the vaccine and specifically in pregnancy. Increasing information on safety has become available, but the basic principles in those podcasts still stand. Podcast on vaccine candidates, podcast on vaccines in pregnancy and lactation, update on vaccines in pregnancy. In May 2021, a large CDC registry of the COVID-19 vaccine in pregnancy was published by Shimabukuro et al. The CDC is tracking pregnant patients who receive the COVID-19 vaccine in 2 ways: (1) the V-safe program (enroll at time of immunization using QR code provided in materials you receive and (2) a pregnancy registry (~5,000 individuals enrolled out of initial V-safe pregnancy population. In the pregnancy registry, the CDC is calling pregnant patients each trimester and post-partum and obtaining records from the obstetric provider. These data have demonstrated no increased risk of pregnancy loss, preterm birth, or congenital anomalies. Of note, many of the pregnancies were not complete at that time, as individuals immunized in the first trimester still had ongoing pregnancies. These data are demonstrated in the graphics below.
The American College of Obstetrics and Gynecology (ACOG) released an updated statement on July 30, 2021 that: recommends that pregnant and lactating individuals be vaccinated against COVID-19. Additionally, the document reinforces that the claims linking COVID-19 vaccines to infertility are unfounded and have no scientific evidence supporting them, recommending vaccination for all eligible people who may consider future pregnancy. Further, although women < 50 years old may receive any COVID-19 vaccine, there is an increased but very rare risk of thrombosis with thrombocytopenia syndrome after the Janssen/Johnson & Johnson vaccine and they may opt for an mRNA vaccine instead (Pfizer or Moderna). The Society for Maternal-Fetal Medicine (SMFM) supports these statements.
A multidisciplinary group at the University of Massachusetts Medical School-Baystate has created decision aids to help pregnant people decide if the COVID-19 vaccine is right for them, which can be found here in 10 languages.




Apple Podcasts , Spotify, Listen Here
The Society for Academic Emergency Medicine has released new Guidelines for Reasonable and Appropriate Care in the Emergency Department (GRACE). These guidelines are aimed at de-implementing low-value care to reduce wasteful medical care. The first guidelines, GRACE-1, target recurrent low-risk chest pain and are published in Academic Emergency Medicine (Musey et al). Previously we covered the American College of Emergency Physician’s clinical policy on non-ST elevation acute coronary syndromes, which does not specifically address recurrent chest pain.
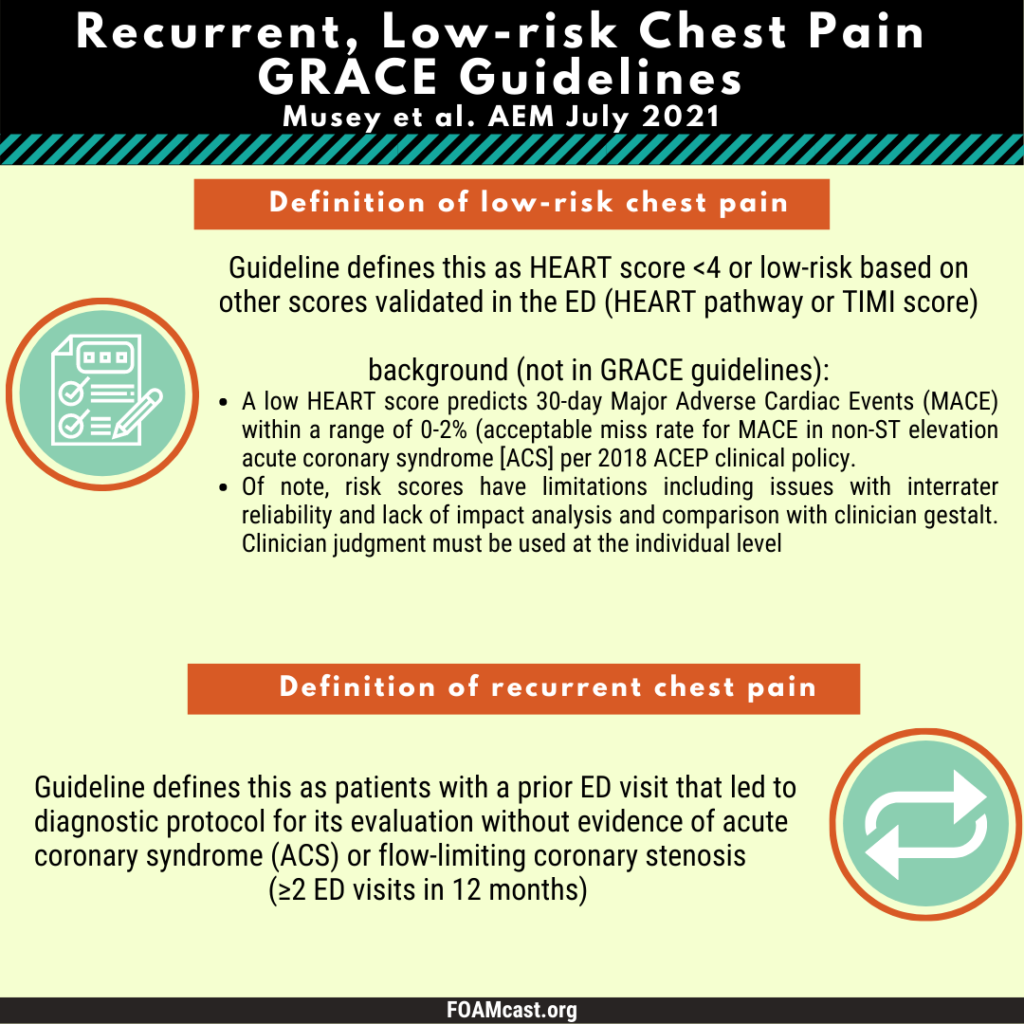


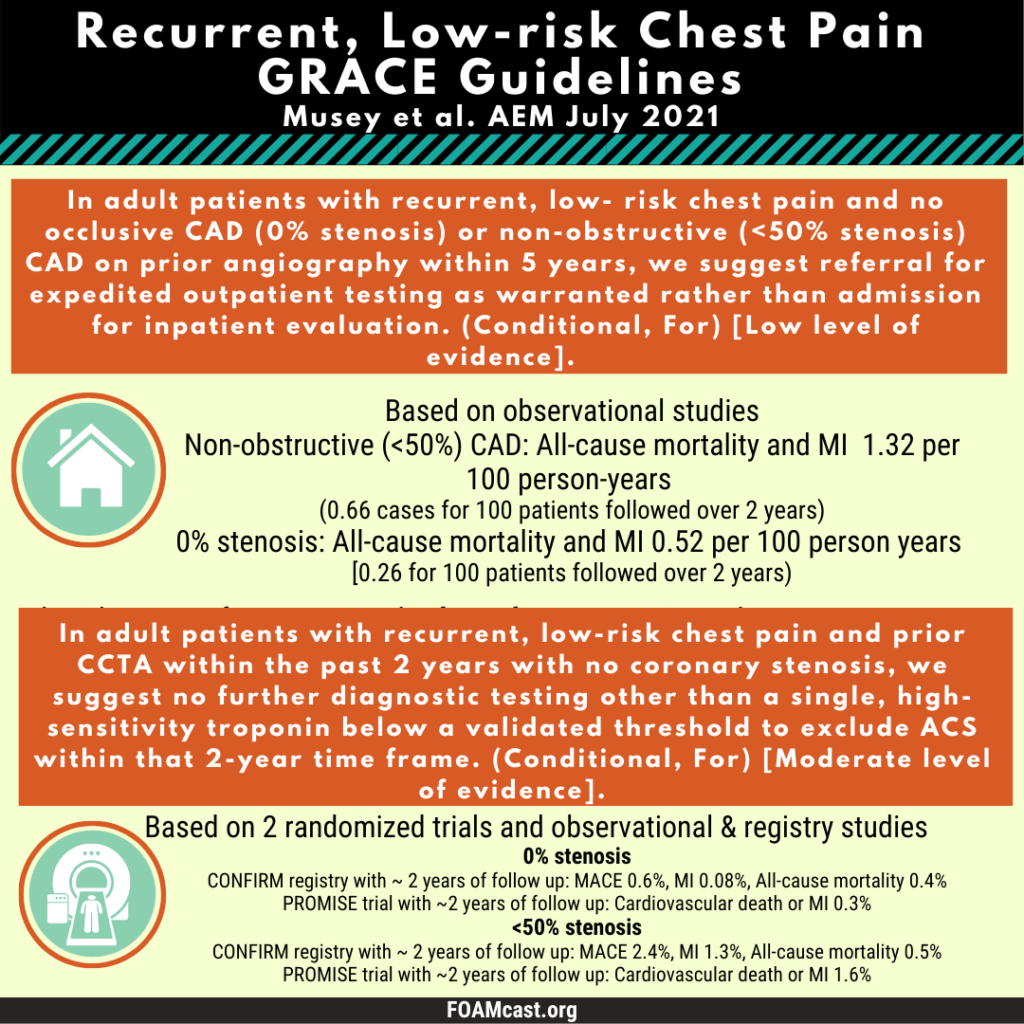

Apple Podcasts , Spotify, Listen Here




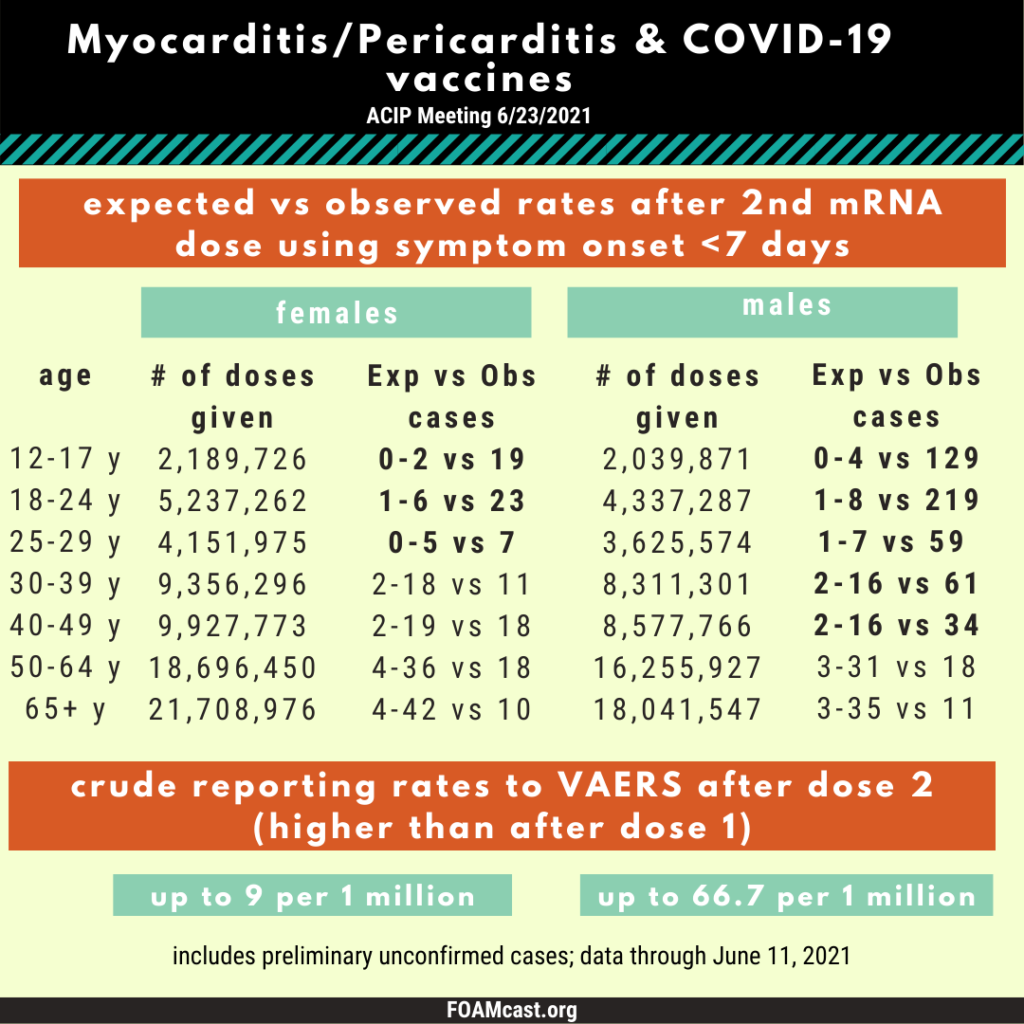
Myocarditis/Pericarditis Cases ACIP Meeting Slides
ACIP Meeting Slides Benefit-Risk Assessment
Myocarditis Data from Israel
Havers et al. Hospitalization of Adolescents Aged 12–17 Years with Laboratory-Confirmed
COVID-19 — COVID-NET, 14 States, March 1, 2020–April 24, 2021 MMWR 2021.
Apple Podcasts , Spotify, Listen Here
Ivermectin is among the potential therapies proposed for COVID-19. In this episode, we review the available randomized trials of ivermectin as well as treatment guidelines.



Apple Podcasts , Spotify, Listen Here
In this episode we cover the literature on tranexamic acid (TXA) in epistaxis, including a new trial in Annals of Emergency Medicine by Reuben et al, the NoPac trial.
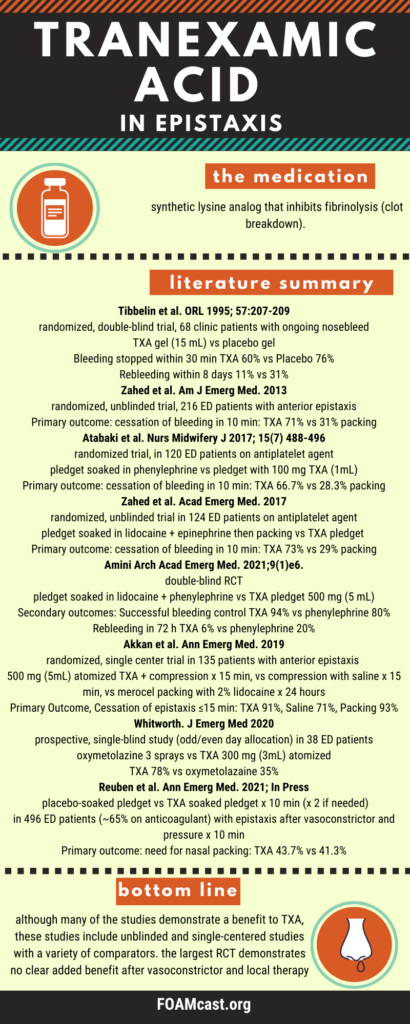
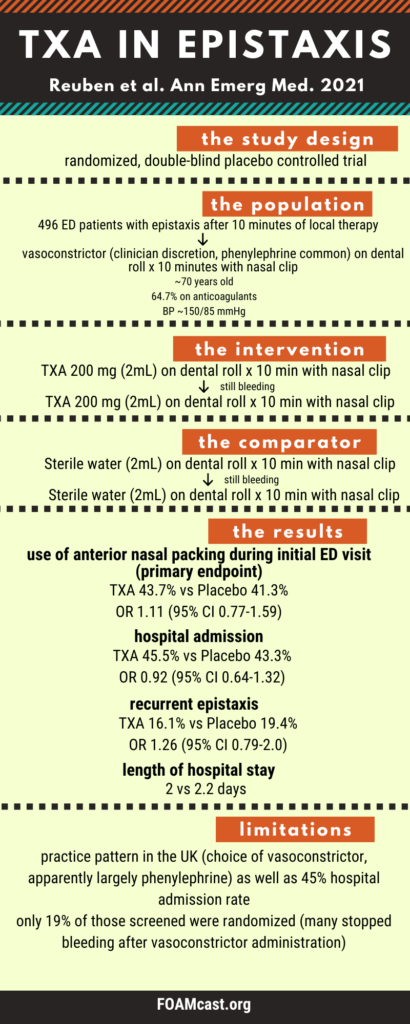
Other references include: